Clinical research is a highly intricate and regulated field that demands meticulous planning, coordination, and execution. The success of a clinical trial hinges on the ability to manage multiple complex tasks, adhere to strict timelines, and maintain the highest standards of quality and compliance. Genelife Clinical Research recognizes these challenges and offers comprehensive Project Management Services tailored to ensure the successful execution of clinical trials, from inception to completion.

Our project management infrastructure is built upon a foundation of highly experienced program and project managers, as well as seasoned investigators. These professionals possess the expertise needed to guide clinical trials from the laboratory to the patient’s bedside with exceptional speed and efficiency.
>Genelife Clinical Research employs well-defined, time-tested SOP that have been honed over years of experience. These SOPs are designed to facilitate the completion of clinical trials within specified timeframes and budgets, enhancing efficiency and minimizing risks.
For clinical endpoint studies, we plan the project resources efficiency and cost-effectiveness without compromising the likelihood of a successful outcome. Our approach is geared toward achieving the best results while optimizing resource utilization. Genelife Clinical Research divides project planning into two major phases. The initial phase involves training and conducting activities like feasibility analysis. After receiving feasibility reports, we develop comprehensive plans covering risk management, communication, quality assurance, safety, monitoring, data management, and statistical analysis. These plans detail how the clinical trial will be conducted within defined stages and with designated resources. They include quantitative objectives, deliverables, risk identification and management, schedule planning, and supporting plans.
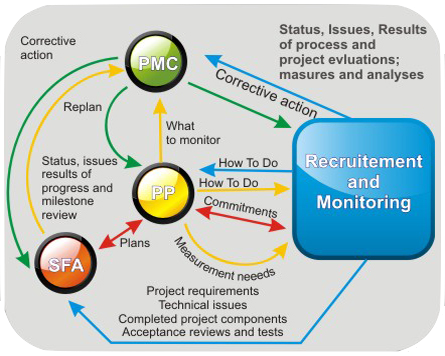
Once a project transitions to the Execution Phase, the project management team ensures that all necessary resources are in place and ready to execute project activities. Activities are delegated to different departments, and progress is closely monitored. Any deviations from the plan trigger appropriate corrective actions, ensuring that the project stays on course.
Project Monitoring and Control is an ongoing process that involves tracking the project’s progress against the plan. This allows us to identify and address deviations promptly. Regular monitoring and control activities are essential for ensuring that the study remains aligned with objectives and timelines.
Effective project management is a cornerstone of study success at Genelife Clinical Research. Our experienced team, well-defined processes, and commitment to therapeutic expertise enable us to efficiently manage clinical trials while maintaining the highest standards of quality and adherence to timelines.
