Genelife Clinical Research Private Limited, with a steadfast Pharmacovigilance department dedicated to the meticulous preparation, review, approval, and reporting of all Serious Adverse Events (SAEs) reported during the course of clinical trials and product development.
The Pharmacovigilance department at Genelife Clinical Research operates on the premise that patient safety is non-negotiable. It is responsible for a myriad of critical functions, including:
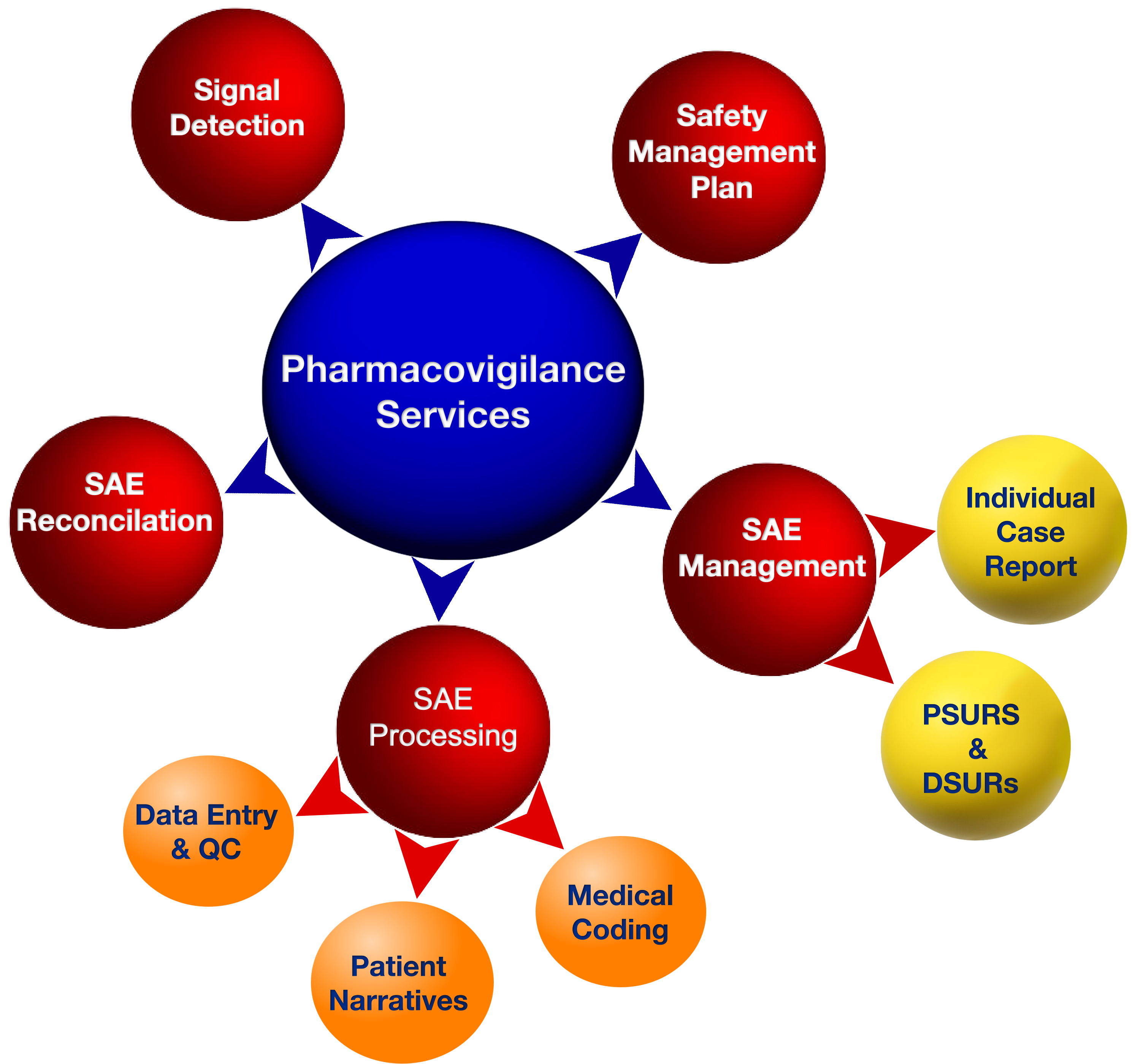
1. AE and SAE Logging, Tracking, and Coding: AEs and SAEs are logged, tracked, and coded meticulously according to recognized standards such as MeDRA/WHOdrug, ensuring data uniformity and accuracy.
2. Safety/Medical Monitoring: A team of experienced safety and medical monitors oversee the safety of patients participating in clinical trials. This proactive monitoring ensures that any emerging concerns are promptly identified and addressed.
3. Medical Review of Data Listings: The Pharmacovigilance team conducts comprehensive reviews of data listings, applying a discerning medical eye to uncover potential safety issues.
4. Review of Medical Coding Data: The accuracy and consistency of medical coding data are paramount. Genelife’s experts meticulously review these codes to ensure adherence to best practices.
5. Preparation of CTD Module II: Genelife excels in the preparation of Common Technical Document (CTD) Module II, a vital component in the drug approval process. It provides a comprehensive overview of the product’s safety and efficacy.
6. Individual Case Safety Reports: Genelife’s Pharmacovigilance team generates Individual Case Safety Reports (ICSRs), compiling detailed information on adverse events and potential links to investigational products.
7. Expedited Reporting of SAEs: Serious Adverse Events demand immediate attention. Genelife expedites the reporting process to ensure that appropriate actions are taken swiftly.
8. Periodic Safety Updates: The department provides periodic safety updates, fostering a proactive approach to safety management.
9. Safety Analysis and Benefit-Risk Evaluation: Safety analyses and benefit-risk evaluations are essential to inform decision-making in clinical trials and product development.
10. Clinical Safety Reports: The creation of comprehensive clinical safety reports underscores Genelife’s commitment to thorough and transparent safety management.
Genelife Clinical Research’s Pharmacovigilance department doesn’t merely report adverse events; it functions as the guardian of patient safety. The department’s coordination of clinical and post-marketing safety data collection, processing, and reporting ensures that potential health risks are identified and mitigated across all stages of clinical trials and product development.
Genelife Clinical Research Private Limited’s Pharmacovigilance department is a symbol of unwavering commitment to patient safety. Their expertise in vigilantly monitoring and reporting AEs and SAEs, paired with their proactive approach, exemplifies their dedication to safeguarding the health and well-being of clinical trial participants. In a field where every data point has the potential to impact lives, Genelife’s Pharmacovigilance department stands as a beacon of diligence and care.
