Clinical operations are a critical component of any successful clinical trial, and at Genelife Clinical Research, we prioritize the meticulous execution of protocols, SOPs, strategies, GCP, and regulations to ensure the timely and successful completion of projects. Our Clinical Operations department is staffed with trained and skilled professionals who are well-versed in the intricacies of clinical trial activities.

Our Clinical Operations team specializes in conducting optimized Pilot, Phase I, Phase II, and III Pivotal ANDA and 505(b)(2) NDA studies. These studies have resulted in successful filings for ANDAs, 505(b)(2), and 505(b)(1) NDAs.
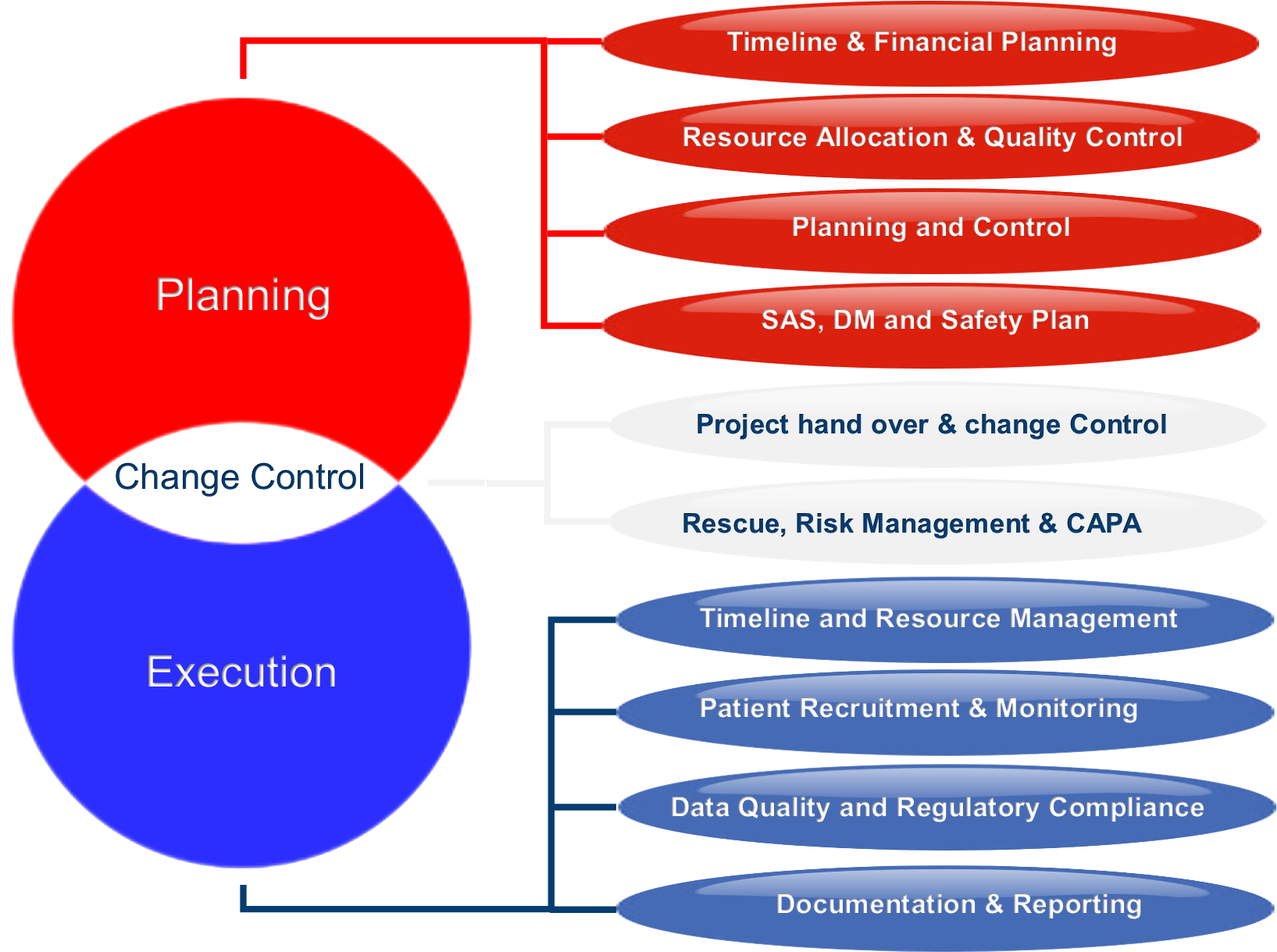
The functions performed by our Clinical Operations department include:
Study Document Management: Ensuring that all study documents are managed efficiently and in compliance with regulatory requirements.
Resource Management and Quality Control: Efficient allocation of resources and rigorous quality control measures to maintain high standards.
Site Feasibility and Selection: Identifying and selecting potential sites, considering factors like patient demographics, disease prevalence, and the track record of investigators.
Monitoring and Control: Overseeing all clinical trial activities and ensuring compliance with protocols and regulations.
Clinical Trial Management System: Utilizing advanced systems for streamlined trial management.
Audits and Regulatory Preparation: Preparing for audits and regulatory inspections to ensure compliance.
Patient Recruitment and Retention Strategies: Developing strategies to recruit and retain study participants effectively.
Worldwide Feasibility Studies: Conducting feasibility studies to assess the viability of trial sites globally.
Tailor-Made Clinical Services: Customizing clinical services to meet the specific needs of each trial.
The Cornerstone of Success
Training and Support: We provide comprehensive training to site personnel, ensuring that they are well-equipped to execute the trial according to the highest standards. Continuous support is offered throughout the trial duration.
Compliance: Genelife’s Clinical Operations teams work closely with sites to ensure strict adherence to local and international guidelines, protocol and SOP.
Patient-Centric Focus: We understand the importance of patient recruitment and retention in clinical trials. Our patient-centric approach ensures that trial participants are well-informed, supported, and valued throughout their participation.
Rapid Subject Recruitment: We have a proven ability to identify and recruit special populations quickly, reducing trial timelines and costs. Our disease surveillance databases contain a vast pool of potential subjects, facilitating expedited recruitment.
Efficient Enrollment Mechanisms: Genelife Clinical Research has established efficient enrollment mechanisms and screening tactics, reducing the overall cost of clinical trials for sponsors. These mechanisms may include referral services, call centers, optimized screening tests sequences, subject retention techniques, and more.
At Genelife Clinical Research, we recognize that the success of clinical trials hinges on the precise execution of clinical operations. Our experienced team and robust processes ensure that trials are conducted efficiently, with a focus on patient safety, data quality, and adherence to timelines and regulations
