Since its inception, Genelife Clinical Research has been at the forefront of transformative advancements in clinical research. Founded in 2010, we have dedicated ourselves to pushing the boundaries of what’s possible in the field of healthcare. Genelife Clinical Research Private Limited embarked on a journey with a clear vision – to bring new treatment options to society. Since our inception, we have been at the forefront of innovation, driving progress in the field of clinical trials for the benefit of our clients and, ultimately, patients in need.
Our foundation is built on the principles of innovation and excellence, aiming to revolutionize the product development process across various domains, including pharmaceuticals, medical devices, biologics, and nutraceuticals. Genelife Clinical Research has consistently introduced groundbreaking methodologies and ideas that have the potential to reshape the industry.
Innovative Approaches for Accelerated Clinical Trials:Fast Patient Recruitment: We have pioneered the use of Disease Surveillance Reports to strategize patient recruitment based on therapeutic areas. This forward-thinking approach enables us to rapidly identify and enroll suitable patients for clinical trials.
High-Quality Data, Low Resource Input: Genelife Clinical Research has developed a working model that ensures high-quality data collection with minimal resource requirements. We believe in delivering excellence without excessive costs.
ThemeCore Lab: As part of our commitment to innovation, we introduced ThemeCore Lab, a unique initiative designed to support clinical trials with core laboratory expertise while keeping expenses in check.

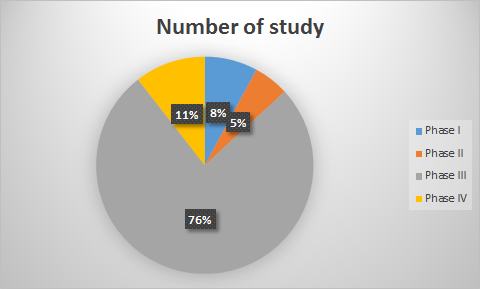
For over 13 remarkable years, Genelife Clinical Research has been on an extraordinary journey, marked by unwavering dedication to advancing healthcare through clinical research. Our rich history encompasses more than a decade of pioneering work, including the successful completion of 55+ clinical studies across various therapeutic areas.
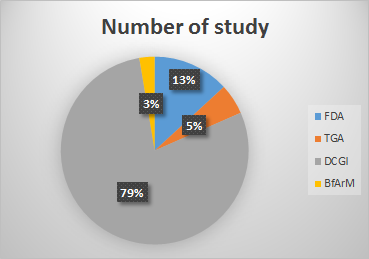
Throughout our journey, Genelife Clinical Research has stood as a beacon of excellence, meeting the stringent standards set forth by renowned regulatory bodies. Our studies have been submitted to and reviewed by esteemed authorities, including the US Food and Drug Administration (FDA), the Therapeutic Goods Administration (TGA), the Federal Institute for Drugs and Medical Devices (BfArM), and the Drug Controller General of India (DCGI).
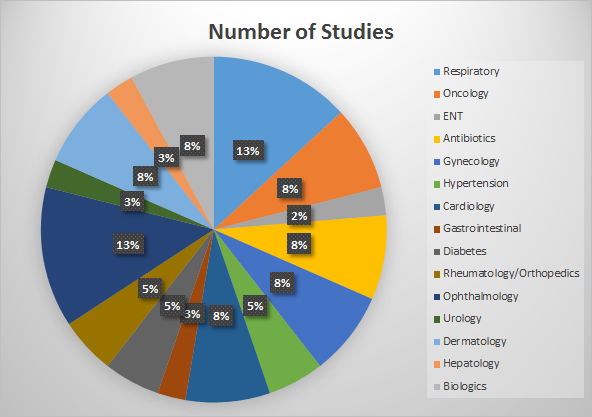
Our expansive portfolio of clinical studies spans diverse therapeutic areas, addressing the unique healthcare needs of patients worldwide. Genelife Clinical Research has been at the forefront of innovation, conducting trials that make a difference in fields such as oncology, cardiovascular health, central nervous system disorders, dermatology, gastroenterology, ophthalmology, and many more.
Throughout our journey, the hallmark of Genelife Clinical Research has been our unwavering commitment to quality and excellence. Our team of dedicated professionals has consistently demonstrated their expertise and dedication, ensuring that every clinical study we undertake adheres to the highest standards.
Our growth journey reflects our ability to deliver results. From a small organization, Genelife Clinical Research has evolved into a respected midsize Clinical Research Organization. This transformation is largely attributed to the word-of-mouth endorsements and loyalty of our clients. We cherish the value of a satisfied client, and in return, we are unwavering in our loyalty to them.